关键区别-平衡方程式与净离子方程式
所有的化学反应都可以写成一个方程式。这个方程的组成部分包括反应物及其物理状态,一个表示反应方向的箭头,以及反应与其物理状态的产物。如果使用了任何特殊条件,它们也会简短地写在箭头上。如果反应处于平衡状态,则在相反方向使用两个半箭头。化学方程式有两种写法:平衡方程式或净离子方程式。平衡方程和净离子方程的关键区别在于,平衡方程反映的是系统中发生的所有反应,而净离子方程只表示特定反应完成后发生的净反应。
内容1。概述和主要区别2。等式3是平衡的。什么是净离子方程式。并列比较-平衡方程式与净离子方程式5。摘要
什么是平衡方程式(a balanced equation)?
Chemical reacti*** are very important in understanding the behavior of a particular system. By writing the correct equation for the reaction, one can get an idea about the changes of different species in that system. For simple reacti*** such as dissolving of NaCl in water, the equation can be written easily by predicting the possible products of that reaction. But for other complex reacti***, further tests may have to be carried out in order to find out the products of that particular system. But most of the times, the written equation is very likely to be unbalanced, which makes it difficult to explain the chemical behavior of that system. Therefore, unbalanced equati*** should also be balanced. The balanced equation includes all reactants and products of that system. The equation is written by c***idering the reactant compound as a molecule. For example, the balanced equation between KI and PbNO3 (if those were reacted in water) would be;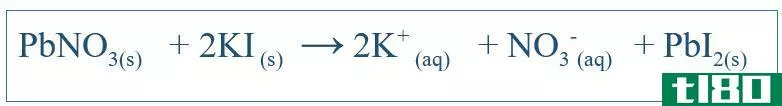
平衡方程的重要性在于,它揭示了应添加的反应物的量,给出了系统中物种之间的化学计量关系的详细信息。在上面的例子中,当向系统中添加这些组件时,PbNO3和KI之间的比率应该是1:2。
什么是净离子方程(a net ionic equation)?
The net ionic equation shows only the overall reaction that has taken place in the system. It includes the ionic species and formed products. But it does not indicate all the reacti*** that have happened in a particular system. For example, if there is a reaction between two molecules using water as the medium, the compounds may have dissolved in water and separated into i***. Some of these i*** may be involved in the reaction but some may not. Then the net ionic equation includes only the i*** that participated in a net reaction. Other i***, which are called spectator i***, are eliminated from the equation to obtain the net ionic equation. For example, if KI and PbNO3 reacted in water, the net ionic reaction would be;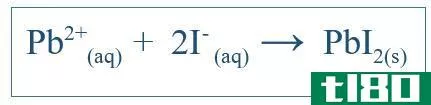
这也应包括K+离子和NO3-离子,但这些离子只会溶解,不参与主要反应;因此,它们不包括在净离子反应中。
平衡方程(balanced equation)和净离子方程(net ionic equation)的区别
| 平衡方程vs净离子方程 | |
| 所有被使用的组分都被写成平衡方程中的反应物。 | 只参与净反应的反应物将写在净离子反应中。 |
| 产品 | |
| 反应结束时的所有组分都包含在一个平衡方程式中。 | 在净离子方程式中,只写净最终产物。 |
| 提供详细信息 | |
| 平衡方程将给出系统中所有物种的详细信息。 | 净离子方程只给出了参与反应的物种的细节 |
总结 - 平衡方程(balanced equation) vs. 净离子方程(net ionic equation)
当某些离子化合物被添加到水中时,它们会分解并形成溶解在水中的离子。这会产生阴离子和阳离子。为了显示反应完成后生成的物种和反应的方向,可以写一个化学方程式。考虑到箭头两侧的每种原子的数量,这个方程可以被平衡;有时,甚至这些物种的电荷也应该考虑在内。这就为那个特定的体系给出了一个平衡的化学方程式。然而,净离子方程只包括反应物。如前所述,平衡方程和净离子方程的关键区别在于,平衡方程表示系统中的所有反应,而净离子方程只显示特定反应完成时发生的净反应。