羟基(hydroxyl)和氢氧化物(hydroxide)的区别
羟基和氢氧化物这两个词听起来非常相似,因为它们都有两个相似的原子,氧(O=16)和氢(H=1)。氢氧化物是一种带单电荷的负离子,羟基不是以自由形式存在的,而是另一分子或离子的一部分。氢氧化离子比分子中的羟基更活泼。这就是羟基和氢氧化物之间的关键区别。
什么是羟基(hydroxyl)?
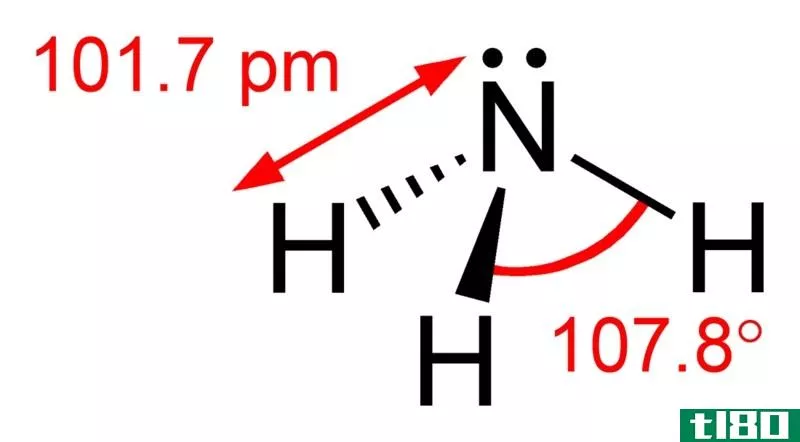
Hydroxyl is a neutral compound and it is the corresponding electrically neutral compound of hydroxide ion. The free form of hydroxyl (•HO) is a radical and when it is bonded covalently to other molecules it is denoted as the hydroxyl (–OH) group. Hydroxyl groups can act as nucleophiles and hydroxyl radical is used as a catalyst in organic chemistry. Hydroxyl groups are not highly reactive as the other nucleophiles. However, they are the facilitators in the formation of strong intramolecular forces called ‘hydrogen bonds’.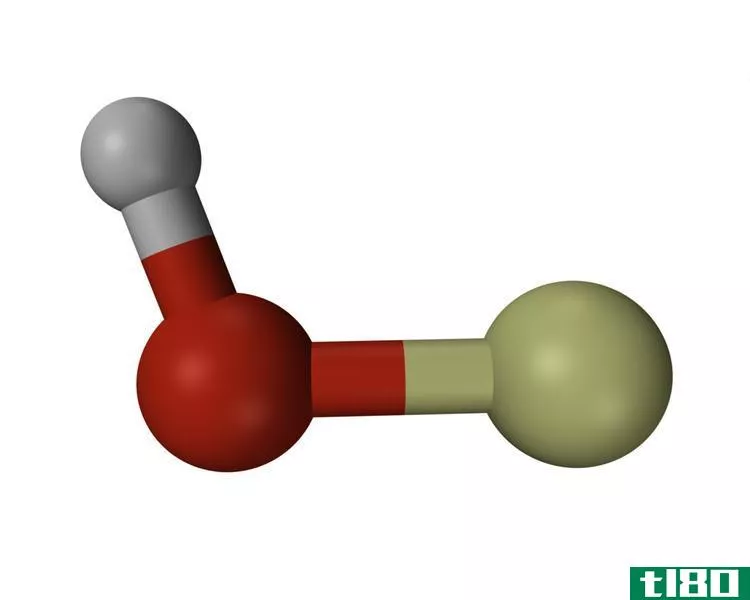
什么是氢氧化物(hydroxide)?
Hydroxide is a diatomic anion which contains an oxygen atom and a hydrogen atom. The bond between oxygen and hydrogen atom is covalent and its chemical formula is OH–. Self-ionization of water produces hydroxyl i*** and therefore hydroxyl i*** are a natural part in the water. Hydroxide i*** are utilized as a base, a ligand, a nucleophile and a catalyst in chemical reacti***. In addition, hydrogen i*** produce salts with metal cati*** and most of them dissociate in aqueous soluti***, releasing solvated hydroxide i***. Many inorganic chemical substances contain the term “hydroxide” in their name, but they are not ionic and they are covalent compounds which contain hydroxyl groups.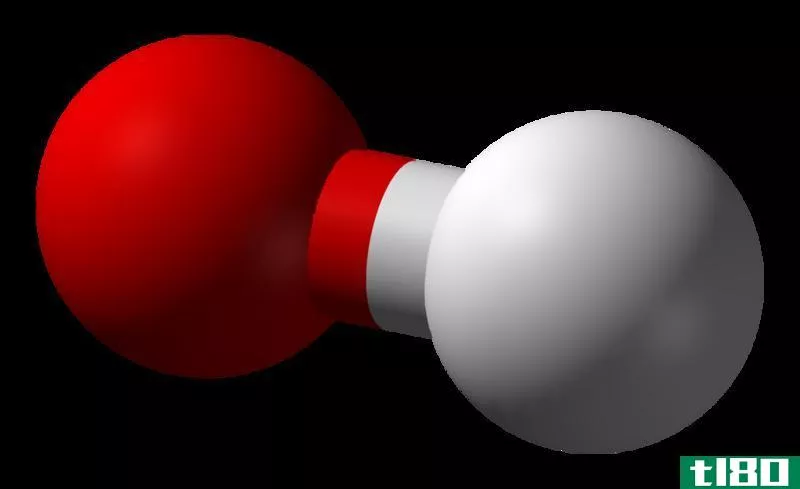
羟基(hydroxyl)和氢氧化物(hydroxide)的区别
结构:
羟基:羟基是一种电中性化合物,有两种形式,自由基和共价结合形式。

羟基自由基与分子共价键合
氢氧化物:氢氧化物是带负电的离子,负电荷在氧上原子属性:
羟基:羟基存在于许多有机化合物中;醇、羧酸和含糖的羟基。含有羟基的化合物,如水、醇和羧酸可以很容易地脱质子化。此外,这些羟基参与氢键的形成。氢键帮助分子粘在一起,这就导致拥有更高的沸点和熔点。一般来说,有机化合物的水溶性较差;当这些分子含有两个或两个以上的羟基时,它们就变得微溶于水。
氢氧化物:大多数含有氢氧化物的化学物质都被认为具有很强的腐蚀性,有些则非常具有腐蚀性有害。什么时候这些化学物质溶于水,氢氧化物离子起着难以置信的强碱作用。由于氢氧化物离子带负电荷,它通常与带正电荷的离子结合。
一些分子中含有氢氧化物基团的离子化合物能很好地溶解在水中;例如氢氧化钠(NaOH)和氢氧化钾(KOH)等腐蚀性碱。然而,其他一些含氢氧化物的离子化合物在水中稍不溶;例如氢氧化铜[Cu(OH)2-亮蓝色]和铁(II)氢氧化物[Fe(OH)2-棕色]。
反应性:
羟基:与羟基相比,羟基的反应性较小。但是,羟基很容易形成氢键,有助于使分子更易溶于水。
然而,羟基自由基具有很高的活性,在有机化学反应中非常有用。
References: