关键区别-乙醇与乙醇酸
虽然乙醇和乙醇酸有相似的名称,但由于它们是两种不同的有机化合物,含有两种不同的官能团,因此可以观察到它们之间的关键区别。乙醇是醇家族中第二简单的成员,而乙醇酸是羧酸族中第二简单的成员。除了分子中存在的官能团外,它们都只含有两个碳原子。当我们比较它们的化学性质时,它们都被用作有机溶剂,并且有一种独特的气味。相反,乙醇酸比乙醇酸。
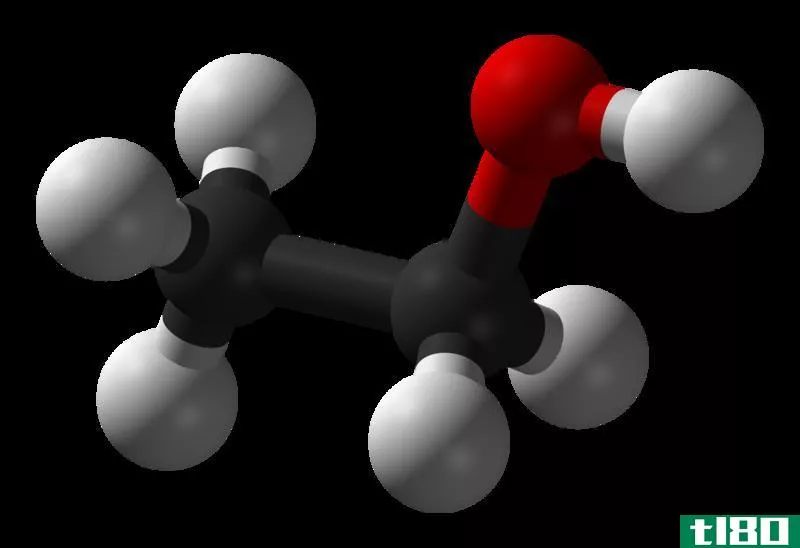
什么是乙醇(ethanol)?
The common name of ethanol is ethyl alcohol. Its functional group is a hydroxyl group (-OH group). All chemical properties such as reactivity, acidity or basicity depend on the functional group. Ethanol has a very mild **ell, and it is a volatile compound. Ethanol is very important in its industrial applicati***; it is a safe solvent, a fuel source, used to produce medicines and co**etics and it is the main component in alcoholic drinks. Ethanol can be produced domestically using agricultural waste such as corn, sugar cane, or grasses.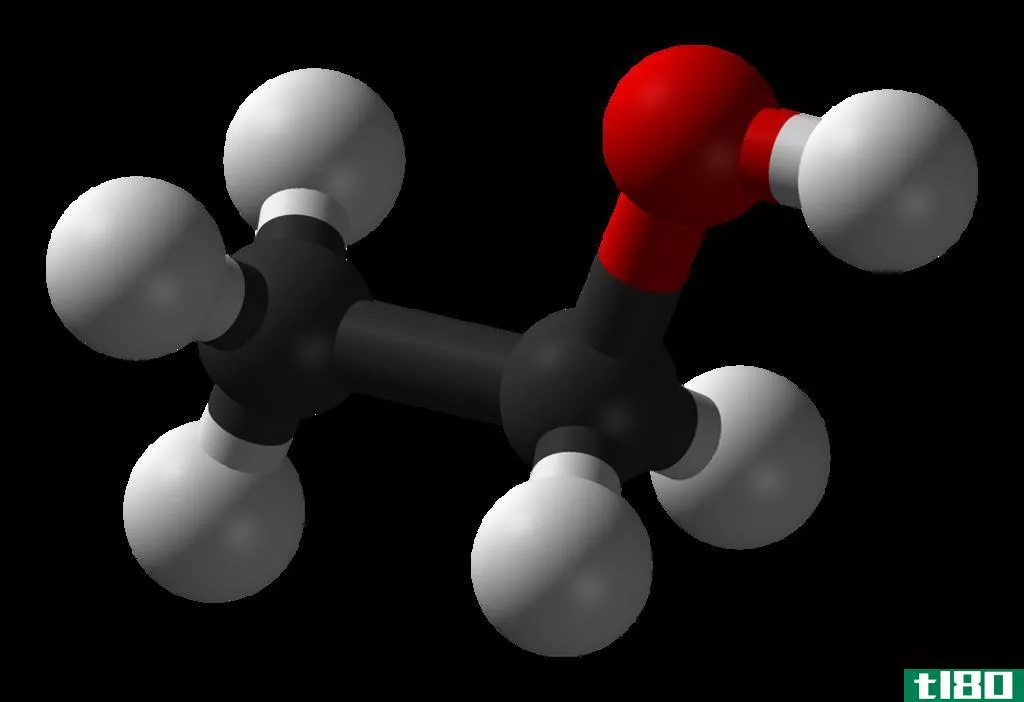
什么是乙醇酸(ethanoic acid)?
The most commonly used name for Ethanoic acid is acetic acid. It is a colourless liquid with a characteristic sour taste and pungent odour. It has the molecular formula of CH3COOH. The undiluted form of the ethanoic acid is called “glacial acetic acid” and roughly about 3-9% of acid by volume is used to make vinegar. Ethanoic acid is c***idered as a weak acid; but, it is corrosive and able to attack the skin.
乙醇(ethanol)和乙醇酸(ethanoic acid)的区别
乙醇和乙醇酸的特性:
功能组:
乙醇:羟基(-OH基)是乙醇中的官能团。这是醇的特性。所有的酒精在其结构中至少有一个-OH基团。
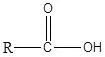
乙醇酸:乙醇酸中的官能团是a–COOH基团。它对所有的羧酸都很常见。
属性:
乙醇:乙醇是一种具有甜味的一元醇,在78.5°C下沸腾。它是唯一能以各种比例溶于水的碳氢化合物。碱性乙醇与酸不反应而生成碱性乙醇。
乙醇酸:在水溶液中为弱酸(pKa=4.76)。液态醋酸是一种极性溶剂,类似于水。它既能溶解极性液体,如糖和盐,也能溶解非极性液体,如油和硫、碘等元素。易与水、氯仿和己烷混溶。醋酸有强烈的刺鼻气味。
使用:
乙醇:酒精存在于酒精饮料中,也用作汽车的生物燃料。许多有机溶剂不溶于水。在涂料工业中,乙醇被用来**许多香水、化妆品和清漆。
乙醇燃料:
CH3CH2OH+3O2→2CO2+3H2O
乙酸:醋酸是一种化学试剂,用来生产其它化学品。大量用于生产醋酸乙烯单体;醋酸乙烯酯可聚合成聚氯乙烯或其他聚合物。此外,醋酸被用来生产酯,用于墨水,绘画和涂料。醋酸酐是另一种重要的化合物,它可以由两个醋酸分子缩合而成。少量的乙醇酸用于生产家用醋。
酸度:
乙醇:乙醇不会与碳酸氢钠(NaHCO₃)发生反应,也不会改变蓝色石蕊纸的颜色。因此,它的酸性比乙醇酸小。
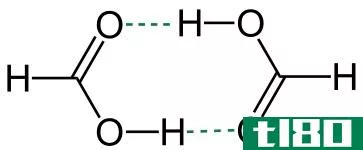
Image Courtesy: “Ethanol-3D-balls”. (Public Domain) via Wikimedia Comm*** “Acetic acid dimer 3D ball” by Jynto (talk) – Own work.(CC0) via Wikimedia Comm***